CSIRO at Work on SOEC Technology
By Stephen H. Crolius on May 21, 2020
Earlier this month the on-line trade journal GasWorld published an interview with Ani Kulkarni, Senior Research Scientist Major Project Leader at Australia’s Commonwealth Scientific and Industrial Research Organisation, that illuminated a research program focused on solid oxide electrolysis technology. The takeaway is that the CSIRO program is making progress that can, in Kulkarni’s words, “elevate this technology from the lab bench to become cost-effective at an industrial scale.”
Solid oxide electrolysis cells are architecturally similar to solid oxide fuel cells, but they differ in that they can also operate in reverse. An SOFC takes a hydrogen-containing fuel and converts it into water and usable electricity. An SOEC can do this as well, but can also go the other way and split water into hydrogen and oxygen. A technology of this nature clearly could play a valuable role in an electricity system characterized by fluctuating loads on the demand side and outputs on the production side that vary with the outputs of renewable generating resources. However, “could play” will never translate into “is playing” if a favorable economic case fails to materialize.
“The focus of our work at CSIRO is to elevate this technology from the lab bench to become cost-effective at an industrial scale,” Kulkarni told GasWorld. The thrust of the research, he said, involves “leveraging materials science and manufacturing technology to improve the electrolyser production process.”
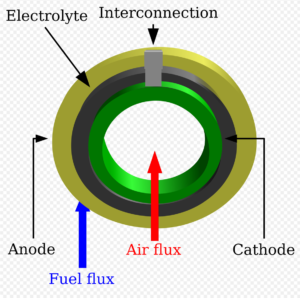
One innovation under exploration by Kulkarni’s team is a tubular cell structure whose shape is better at managing thermal flux than the flat plates found in conventional fuel cell designs. The “tubular cell structure … vastly reduces the problems of thermal stress, and the electrodes will have a significantly longer life and lower maintenance costs,” Kulkarni said. He continued:
In operation, our tubular cell structure also has advantages. Since the heat dissipation is better, we can ramp up the electrolyser in less than 30 minutes. This is much faster than can be achieved with a flat stack structure. That improved dynamic response means the SOE system can be used effectively in transient power management schemes to convert excess electrical production to hydrogen.
Ani Kulkarni, quoted in CSIRO exclusive: Solid oxide electrolysis growing on an industrial scale, Gasworld, May 4, 2020
Kulkarni was co-author of a 2018 paper published in the technical journal Electrochimica Acta (“Evaluation of Sc2O3–CeO2–ZrO2 electrolyte-based tubular fuel cells using activated charcoal and hydrogen fuels”) that detailed the characteristics of an SOFC employing tubular cells made of the same materials as those under investigation for the SOEC.
The CSIRO research is also addressing electrode materials and production techniques. Among other innovations, Kulkarni’s team has developed a material that can be used for both anodes and cathodes. “In current SOE systems,” Kulkarni said, “the anode and cathode are different compositions. The benefit of using the same material on the anode and cathode is that the electrodes can be manufactured in a single step.”
A March 2019 Ammonia Energy article describes parallel work on SOECs by Danish engineering and technology development company Haldor Topsoe. In this case, the focus is on deploying an SOEC as the hydrogen generation unit within an ammonia production train. Here, too, the ultimate goal is to establish the SOEC as a technology with realistic commercial prospects, although Haldor Topsoe’s focus is on using an SOEC to enhance the energy efficiency of the overall ammonia production process. One deliverable from the current phase of the company’s work, which is scheduled to run through 2022, is a feasibility study for a small industrial-scale green ammonia plant. If all goes well, the plant itself could be operational by 2025 and the technology could be commercially available by 2030.
Although ammonia production is not within the scope of his team’s work, Kulkarni sees the nexus between SOEC technology and ammonia as a hydrogen carrier. On the one hand, he said, “Here in Australia, we have many locations that are ideally placed to harvest solar energy to produce green hydrogen which can be used in the domestic economy or exported to Asian markets as liquid hydrogen, methanol or ammonia.” And “on the other hand, the long-term storage of ammonia or methanol does not result in significant boil-off losses so SOE opens up the possibility of using a wider range of hydrogen carriers in fuel cells.”
The CSIRO research team includes participants from Australia, Israel, the United Kingdom, and the United States.