New R&D: ammonia as a green steel enabler
By Julian Atchison on April 10, 2023
Reductant for steelmaking
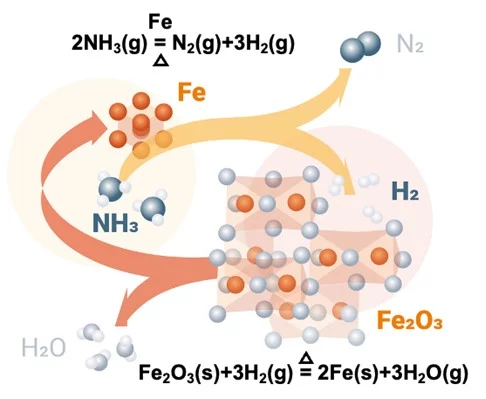
Researchers at the Max Planck Institute for Iron Research have released new research on the direct use of ammonia in steelmaking. In a laboratory-scale reactor, ammonia was successfully used as a reductant to convert iron ore to sponge iron. Compared to a hydrogen-based reduction reaction, the ammonia-based experiment demonstrated similar kinetic characteristics, and yielded the same “metallization degree”. The overall reaction is driven by the decomposition of ammonia to hydrogen at elevated temperatures (700°C) in the presence of porous iron, with hydrogen subsequently reducing the iron ore.
Researchers note that the formation of nitrides in the reactor formed a protective phase over the sponge iron (one which is subsequently removed in processing the sponge iron), and that by-product nitrogen could act as a “heat carrier” to help maintain reaction temperature in an industrial furnace.
The next step for the Max Planck team will be adjusting different parameters, speeding up the reaction to what is required in an industrial setting. The research is supported by an Advanced Grant of the European Research Council for Prof. Dierk Raabe, director at MPIE, and by a grant for Dr. Yan Ma financed by the Walter Benjamin Programme.
Avoid cracking, save energy
The use of hydrogen as a direct reductant for iron ore has already been demonstrated, and shows promise globally. But as the team at Max Planck argues, the direct use of ammonia takes advantage of existing global infrastructure for ammonia transport & avoids a suite of extra costs. By removing the need for either hydrogen transport & storage or cracking ammonia on arrival, cheaper and more energetically efficient steel is produced.
ADR (ammonia-based direct reduction) is kinetically as effective for producing green iron as HyDR at 700 °C. The direct utilization of ammonia in the reduction process offers a process shortcut, alleviating the need for a preliminary ammonia cracking step…[It] provides a novel approach to deploying intermittent renewable energy for an unprecedented and disruptive technology transition toward sustainable metallurgical processes. With these benefits, it connects two of the currently most greenhouse gas intense industries (namely, steel and ammonia production industries) and opens a pathway to render them more environmentally benign and sustainable.
Conclusion, from Ma et al., Reducing Iron Oxide with Ammonia: A Sustainable Path to Green Steel (Adv. Sci., Mar 2023)