The Ammonia Academic Wrap: a new breakthrough for eNRR research and more
By Julian Atchison on June 29, 2021
Welcome to the Ammonia Academic Wrap: a summary of all the latest papers, developments and emerging trends in the world of ammonia energy R&D.
New breakthrough for eNRR research
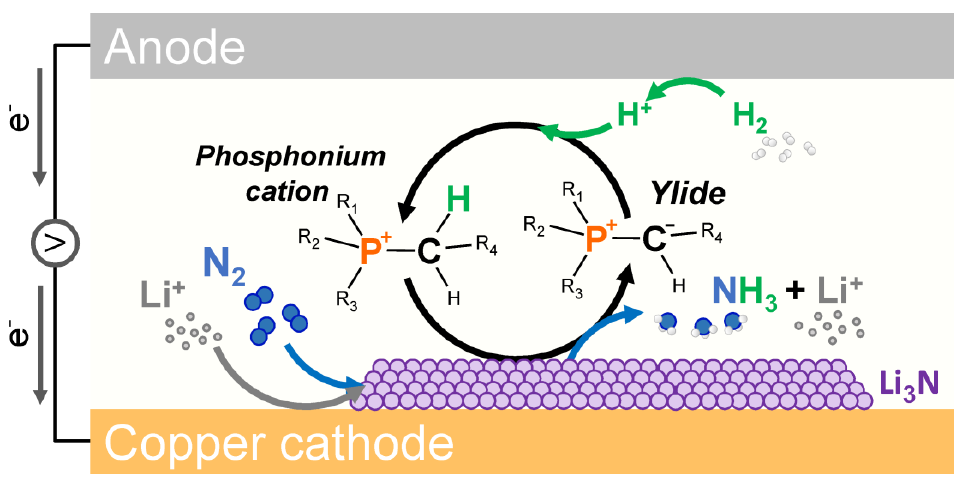
Electrochemical nitrogen reduction (eNRR) to produce ammonia is not a new concept, but to date researchers have been confronted with two main issues: low production rates (typically two orders of magnitude below that required for any industrial applications), and the issue of “sacrificial” proton sources which are continuously depleted in the reaction cell (and which must be carefully replenished, or else the reaction balance swings towards hydrogen production). In their new Science paper, a team at Monash University presents a possible new way forward via the use of a phosphonium salt as a non-depleting proton source. They achieve a sustained eNRR production rate better than any previously reported, along with a much-improved Faraday efficiency, stable cell voltage and unblemished electrode working surface. Though the achieved nmol s-1 cm-2 rate is still well below that required for industrial applications, Professor Doug Macfarlane is confident this result is a key milestone on the road to commercialising the next generation of ammonia synthesis technology. Learn more about this research and hear some extra thoughts from Doug in the June episode of Ammonia Energy Live.
Ammonia production from food waste and brown-water
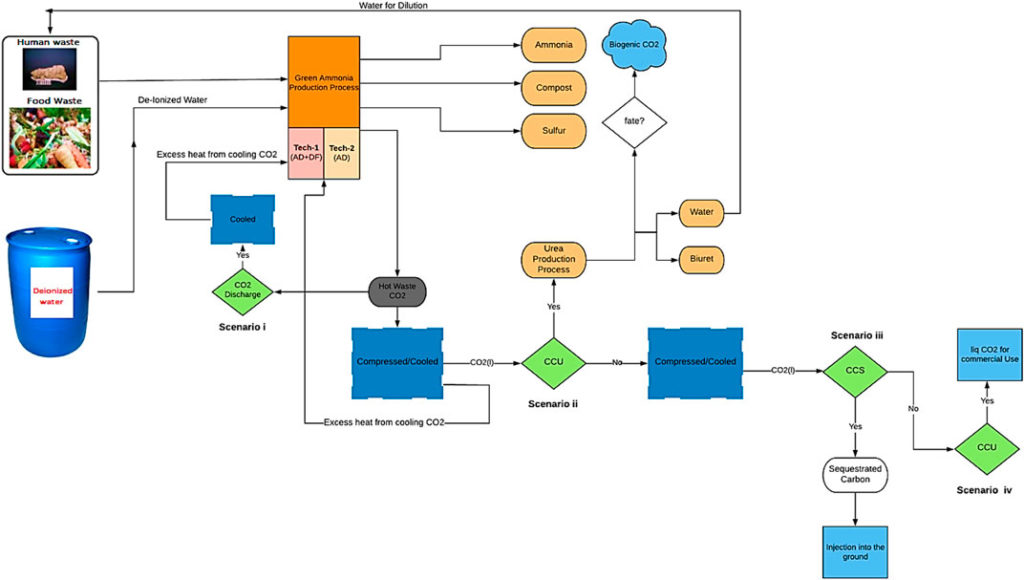
Researchers at the University of Sheffield estimate significant potential energy and emissions savings are possible by producing feedstock hydrogen from waste (either food waste or brown water/human waste), compared to current industrial processes (electrolysis and steam methane reforming). In their recent Frontiers in Energy Research paper, they concluded the most efficient modeled setups combined a 2-stage, dark fermentation of waste coupled with membrane separation of fermentation product gases, CCS or CCU (liquid carbon dioxide capture and underground storage or utilisation off-site), a SOFC for power generation and a standard Haber-Bosch (HB) reactor. Overall, this produced ammonia with 41% less energy consumption than electrolysis-HB, and ~30% less energy consumption than SMR-HB.
Huge potential of green ammonia production from hybrid solar-wind across the globe
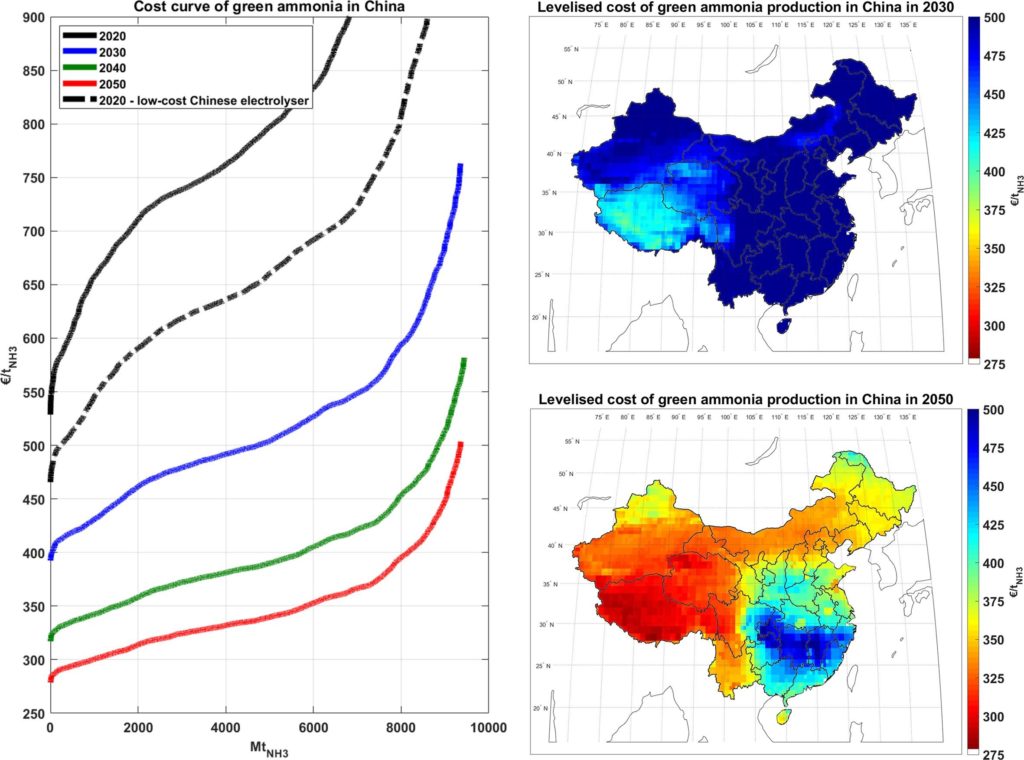
In their new Applied Energy paper, a quartet of Finnish researchers have landed on some eye-popping numbers. Green ammonia can be produced for 370–450 €/tonne in all continents by 2030. By 2050, those production costs could decline to 285–350 €/tonne at the best sites (the Patagonia and Atacama deserts in South America show the best solar-wind hybrid potential, according to the authors). China also shows huge potential: by 2040, green ammonia in China would be lower in cost than ammonia from new coal-based plants, even at the lowest coal prices and no “greenhouse gas emissions cost”. The authors conclude that for “greenhouse gas emissions cost” of just 75 €/tCO2, green ammonia in 2040 would be cost-competitive even if natural gas at prices are as low as 2 USD/MMBtu, bringing the cost crossover point between fossil and renewables-produced ammonia even closer.
Predicted cost dynamics of electrolyser technology
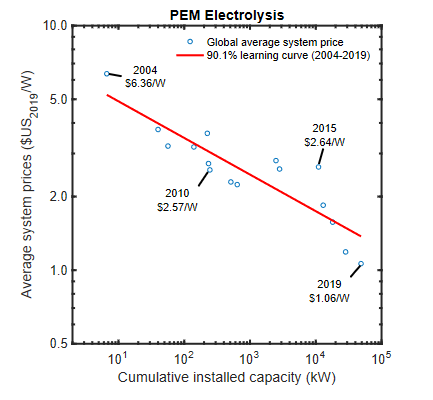
In their new open-access SSRN paper, researchers at the University of Mannheim use Theodore P. Wright’s classical “learning by doing” approach to assess future cost dynamics for PEM electrolyser technology. This approach (first used in 1936) predicts that cost will fall as a function of the cumulative volume of past deployments, and has held true for a series of industries and processes for nearly a century – with clean energy being no exception! Using historical data for solar PV, on and offshore wind, lithium-ion battery and electrolyser technology, the authors show that each time deployment doubles, costs drop by between 9 and 39% (the accepted “benchmark” is 20%). Although electrolysers are only progressing at a 9% rate, the authors suggest: i) “interdependent learning effects” between green hydrogen and other clean technologies will likely lift this figure, and ii) policymakers (particularly in the US) recognise this slower progress and will look to intervene with incentives for electrolyser deployment.
Hydrogen production using selective ion membranes
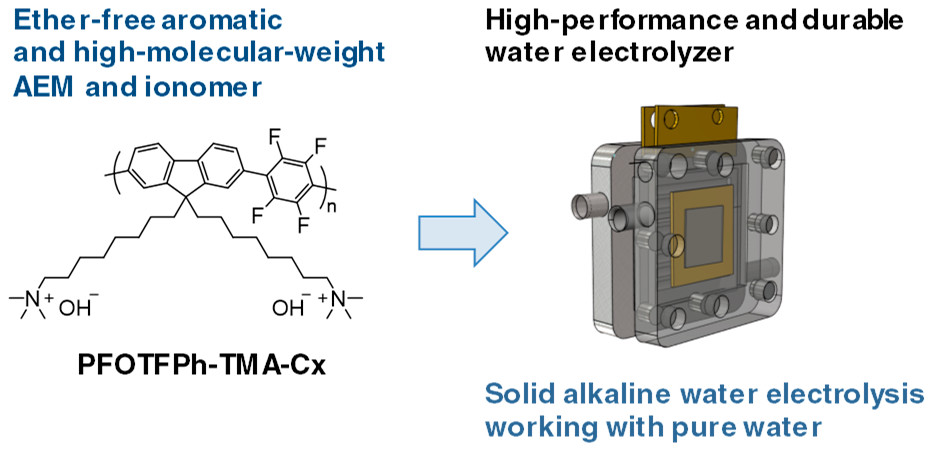
A team at the Tokyo Institute of Science has demonstrated a breakthrough in research towards solid alkaline water electrolysis or SAW – seen as a potential alternative to PEM electrolysis. Where PEM uses costly, rare materials for its membrane technology, SAW utilises inexpensive anion exchange membranes, though to date progress has stalled because these cheaper membranes tend to be less durable and often unstable. In their recent Applied Energy Materials paper, researchers demonstrated sustained pure water electrolysis at 80°C using a membrane based on fully aromatic and high-molecular-weight poly(fluorene-alt-tetrafluorophenylene) modified with trimethylammonium.
Get the Ammonia Wrap straight to your inbox every week
Make sure you’re signed up for AEA email updates, including our weekly newsletter featuring wrap articles just like this one.