Process Superstructures and the Production of Cost-Advantaged Ammonia
By Stephen H. Crolius on February 01, 2018
At the 2017 NH3 Energy+ Topical Conference, graduate student Doga Demirhan reported on an ongoing investigation at the Energy Institute at Texas A&M University. The work involved evaluation of options for an ammonia production system and concluded that biomass could be an economically viable feed stock under current, real-world conditions. This is a notable outcome. Just as notable is how it was reached.
Everyone knows the basic principles of business strategy. Commodity prices are set by markets. Cost positions are achieved by individual suppliers based on their particular configuration of production elements. A supplier’s profit is the difference between the market price and his individual cost. Superior profits accrue to suppliers with low costs relative to their competitors. Capital flows to opportunities that are based on advantaged cost positions.
Identifying situations that could yield advantaged cost positions is a challenging endeavor, yet a critical one for the economic implementation of ammonia energy. A number of presentations at the Topical Conference spoke to how economic implementation could be best achieved. Often referenced was the fundamental parameter of cost per unit of material or energy. Calculations were shown, quantitative outcomes shared, and indications of advantage for ammonia demonstrated in a dozen scenarios.
But the Texas A&M team addressed the topic with a particularly ambitious methodology in their paper, Process Synthesis and Global Optimization of Novel Ammonia Production Processes. The approach in question has gained currency in the broader chemical industry in recent years. It was summarized in the Texas A&M abstract in the following terms: “… we propose the development of an ammonia process superstructure that not only captures the traditional Haber-Bosch process, but also incorporates renewable energy sources and alternative ammonia production routes.”
The key term here, “process superstructure,” refers to a data set that comprehends the range of parameters associated with a chemical production process and that organizes those parameters so that potential configurations of the production process can be evaluated mathematically.
The Texas A&M abstract continues, “Through this optimization-based process synthesis, we can extract the optimal ammonia plant topology under different production scenarios. All process alternatives will be rigorously modeled and incorporated into the superstructure.”
If it sounds like this could involve serious number-crunching by a heavyweight computer, that’s because it does. As the abstract says, “The overall problem is a large-scale nonconvex mixed-integer nonlinear optimization (MINLP) model, which is solved to global optimality using a deterministic branch-and-bound algorithm.”
The Texas A&M Energy Institute is a major center of energy industry research whose “charter partner” is Royal Dutch Shell. Its 250 faculty affiliates are active across 65 research topics ranging from Petroleum Exploration and Production to Nuclear Energy Conversion Technologies to Smart Grid Architecture to Materials for Wind Energy. It also houses the Multi-Scale Systems Engineering Laboratory (MSEL) which provides access to high-performance computing systems that can take on MINLP optimization problems.
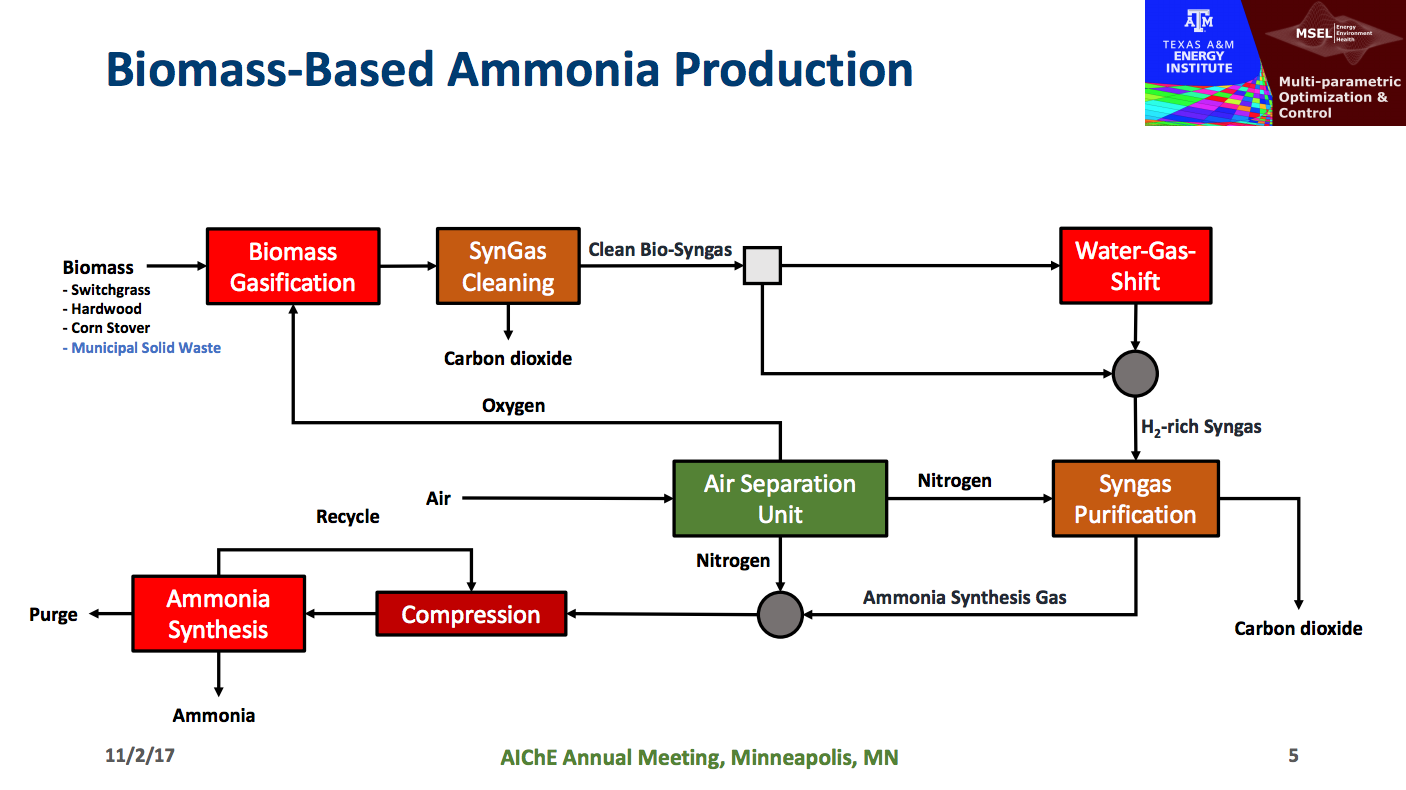
The Texas A&M team set up the exercise such that “feed stock options will include renewables (e.g. biomass, municipal solid waste, etc.) and fossil fuels (e.g. natural gas, coal). Although the feed stocks under consideration are all hydrocarbons, wind and solar energy will also be considered to supply the energy requirements for the process.” Demirhan’s presentation focused on a specific comparison between ammonia production processes based on natural gas and those based on gasification of biomass. Model elements included mass-and-energy-balance equations; thermodynamic relations including phase and chemical reaction equilibria; parameters for simultaneous heat and power integration; and “concave cost functions for calculation of unit investment costs.”
When the computational wheels stopped spinning, the team’s conclusion was that for plants of the right scale, “ammonia produced from hardwood type biomass had the lowest break-even price,” i.e., a lower breakeven price than that for a plant using natural gas feed stock. Note that the plant with the lowest breakeven price per ton by definition has the lowest cost.
The way in which the MINLP optimization methodology can help move the ammonia energy agenda forward was also illustrated by the another team that presented at the conference, this one under the leadership of Prodromos Daoutidis at the University of Minnesota. Although the presentation itself, Dynamic Modeling and Optimization of an Absorption Enhanced Ammonia Synthesis Process, focused on a technical improvement in the ammonia separation step of the production process, the team is working within the same system optimization framework as the Texas A&M group.
In June 2016 Daoutidis published the results of an MINLP analysis whose key focus was the potential investment case for relatively small-scale “distributed” ammonia plants in Minnesota. His paper in the Proceedings of the 26th European Symposium on Computer Aided Process Engineering, Ammonia supply chains: A new framework for renewable generation with a case study for Minnesota, was based on a model consisting of “2,518 decision variables … 150 constraint equations [and] 27 nonlinear elements.” It ultimately showed that the global cost of ammonia fertilizer supply to Minnesota farmers could be reduced to its minimum level by the addition of a single plant within the state’s boundaries. It specified both the location and scale of the plant, which would be relatively small, at 140,000 tons per year, and would supply 27% of the state’s annual ammonia demand.
A PDF version of Demirhan’s conference presentation is available to download at the NH3 Fuel Association website. We look forward to reporting on the next round of results from this ongoing program at Texas A&M University as well as from the University of Minnesota in the coming months.